Global Journal of Medical and Clinical Case Reports
Pharmacology of Senolytics for Lung Aging and COPD: An Emerging Therapeutic Frontier
1Assistant Professor, Faculty of Pharmacy, Noble University, Junagadh, India
2Professor, Faculty of Pharmacy, Noble University, Junagadh, India
3Dean, Dr. Subhash Ayurveda Research Institute, Dr. Subhash University, India
Author and article information
Cite this as
Sarah Gadavala F, Buddhadev S, Buddhadev S. Pharmacology of Senolytics for Lung Aging and COPD: An Emerging Therapeutic Frontier. Glob J Medical Clin Case Rep. 2026:13(2):026-031. Available from: 10.17352/gjmccr.000241
Copyright License
© 2026 Sarah Gadavala F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Abstract
Chronic obstructive pulmonary disease is strongly influenced by accelerated lung aging and an accumulation of senescent cells within airway epithelial and immune compartments. Senescent cells release a spectrum of inflammatory mediators that worsen tissue destruction and impair repair mechanisms. Conventional COPD therapeutics do not adequately address cellular senescence, which has led to growing interest in senolytic drugs that selectively eliminate senescent cells or reduce harmful secretory signaling. Several senolytic candidates, including Dasatinib–Quercetin, Fisetin, Navitoclax, HSP90 inhibitors, and peptide-based approaches, have demonstrated promising effects in preclinical respiratory models. These agents modulate pathways such as BCL-2 inhibition, suppression of PI3K/AKT signaling, activation of mitochondrial apoptosis, or disruption of FOXO4–p53 interactions. Early studies suggest potential for reducing chronic inflammation, restoring epithelial function, and slowing structural deterioration in the lung. Although human evidence remains limited and concerns surrounding toxicity, delivery, and biomarker development persist, senolytics represent an emerging pharmacological avenue for targeting lung aging and may hold therapeutic value in COPD.
Introduction
Chronic obstructive pulmonary disease continues to pose a major global health concern due to progressive airflow obstruction, chronic inflammation, and irreversible damage to lung architecture [1]. Although several risk factors, including tobacco smoke and environmental pollutants, have been extensively studied, the contribution of accelerated lung aging has gained considerable attention in recent years [2]. Aging lungs exhibit impaired tissue repair, heightened oxidative stress, and accumulation of senescent cells, all of which contribute to pathophysiological features of COPD.
Senescence is characterized by permanent cell-cycle arrest accompanied by metabolic activity and release of proinflammatory mediators. These senescent cells accumulate in airway epithelium, fibroblasts, endothelial cells, and immune cell populations of COPD patients, amplifying chronic inflammation and promoting tissue degradation through the senescence-associated secretory phenotype [3]. Conventional COPD therapy mainly focuses on bronchodilation and anti-inflammatory actions, but does not eliminate senescent cells. As a result, senolytics have emerged as a novel pharmacological class aimed at selectively removing senescent cells or modulating senescence-associated pathways [4].
This mini-review summarizes the concept of cellular senescence in lung aging, outlines the mechanisms of established and emerging senolytic agents, and highlights their potential relevance for COPD management.
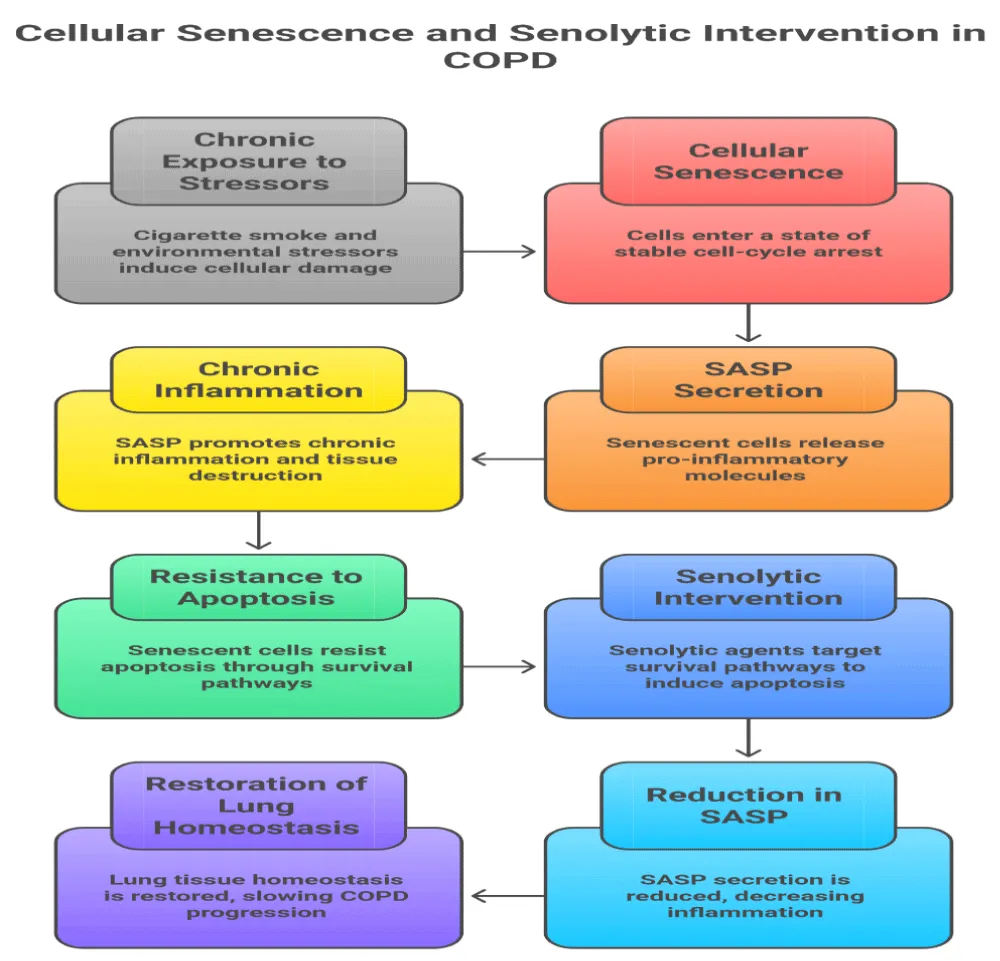
The mechanistic interplay between lung aging, cellular senescence, SASP release, and the therapeutic action of senolytic agents in COPD is illustrated in Figure 1. This schematic summarizes the key molecular pathways driving disease progression and highlights potential pharmacological targets for senolytic intervention.
Biology of cellular senescence in lung aging
Senescence arises in response to persistent genomic, oxidative, or environmental stress. Telomere shortening, mitochondrial impairment, and chronic exposure to cigarette smoke promote the transformation of normal cells into senescent phenotypes [5]. Once established, these cells exhibit stable growth arrest mediated by upregulation of cyclin-dependent kinase inhibitors such as p16INK4a and p21Cip1 [6].
A key feature of senescence is the senescence-associated secretory phenotype. This phenotype encompasses secretion of cytokines such as IL-6, IL-8, TNF-α, matrix metalloproteinases, chemokines, and growth factors [7]. In the lung, SASP contributes to persistent inflammation, recruitment of immune cells, apoptosis resistance, mucus hypersecretion, and extracellular matrix degradation. This environment accelerates lung aging and exacerbates structural damage characteristic of COPD [8].
Senescent cells also impair regenerative capacity by occupying niches needed for healthy epithelial and endothelial renewal. Markers such as SA-β-galactosidase, CDKN2A, and DNA damage foci are commonly detected in lung tissues of COPD models [9]. The accumulation of these cells offers a mechanistic explanation for the chronic, non-resolving nature of COPD.
Senolytics: A new pharmacological class
Senolytics are defined as agents that selectively induce the death of senescent cells, sparing non-senescent cells by exploiting vulnerabilities in survival pathways [10]. Senescent cells depend on anti-apoptotic networks including BCL-2 family proteins, PI3K/AKT signaling, and p53 interactions, making these pathways attractive targets for pharmacological intervention.
Another related category is Senomorphics, which do not kill senescent cells but attenuate harmful SASP signaling. Senomorphics, including metformin and rapamycin, help reduce inflammatory mediator release and may complement true senolytic therapy [11].
Senolytics may benefit COPD by reducing the senescence burden, decreasing chronic inflammation, restoring epithelial function, and slowing the progression of emphysematous changes. Preclinical evidence has shown improvements in lung compliance and reductions in inflammatory infiltration following senolytic exposure [12].
Pharmacology of key senolytic agents for lung aging
Dasatinib and Quercetin
A widely studied senolytic combination consists of Dasati nib, a tyrosine kinase inhibitor, and Quercetin, a flavonoid with antioxidant and anti-inflammatory activity. Together, they target senescent-cell anti-apoptotic pathways, including BCL-2 family proteins and PI3K/AKT signaling, leading to selective apoptosis [13]. In respiratory models, Dasatinib–Quercetin reduces inflammatory cytokine production and improves tissue structure by clearing senescent epithelial and endothelial cells [14]. Pharmacokinetically, Dasatinib has good systemic absorption, whereas Quercetin suffers from limited bioavailability, prompting interest in improved formulations. Toxicity remains low when administered intermittently, which is an advantage in chronic diseases [15].
Fisetin
Fisetin is a naturally occurring flavonoid with strong senolytic potential. It influences multiple pathways, including reduction of SASP mediators, enhancement of Nrf2 antioxidant signaling, and modulation of mitochondrial function [16]. Unlike synthetic agents, Fisetin shows a favorable safety profile and has been evaluated in preclinical models of lung inflammation. Its senolytic activity appears to be cell-type dependent but robust in epithelial systems relevant to COPD [17].
Navitoclax [ABT-263]
Navitoclax is a potent inhibitor of BCL-2 and BCL-XL, two key anti-apoptotic proteins upregulated in senescent cells. By impairing these survival pathways, Navitoclax induces mitochondrial-dependent apoptosis [18]. In lung studies, Navitoclax effectively clears senescent fibroblasts and epithelial cells, reducing SASP and improving structural parameters [19]. However, its clinical utility is limited due to dose-dependent thrombocytopenia, as platelets also rely on BCL-XL for survival [20]. Intermittent or targeted pulmonary delivery may mitigate these concerns.
FOXO4–DRI peptide
FOXO4–DRI is a synthetic peptide designed to interfere with the FOXO4–p53 interaction, a mechanism that helps senescent cells resist apoptosis. Disruption of this interaction releases p53, initiating programmed cell death [21]. Although research on its respiratory applications is still early, FOXO4–DRI has shown effectiveness in clearing senescent epithelial cells. Its short half-life and peptide nature raise the possibility of inhalation-based delivery for lung diseases [22].
Metformin and Rapamycin [Senomorphics]
Although not true senolytics, both agents modify SASP expression and can reduce senescence-induced inflammation. Metformin activates AMPK, decreases oxidative stress, and modulates metabolic pathways relevant to aging [23]. Rapamycin, through mTOR inhibition, improves autophagy and reduces inflammatory signaling. Both compounds have shown protective roles against cigarette-smoke–induced senescence in preclinical COPD models [24].
Selectivity and senescence-specific vulnerabilities
A defining characteristic of senolytics is their preferential targeting of senescent cells, which rely disproportionately on pro-survival networks termed senescence-associated anti-apoptotic pathways [SCAPs]. These include BCL-2/BCL-XL overexpression, PI3K/AKT hyperactivation, and altered p53 regulation [25].
Senescent cells display mitochondrial dysfunction, increased ROS production, and metabolic reprogramming, rendering them more susceptible to mitochondrial apoptosis triggers compared with proliferating cells. However, this selectivity is not absolute. For example, Navitoclax-induced thrombocytopenia illustrates that non-senescent platelets also depend on BCL-XL.
Potential resistance mechanisms may include:
- Upregulation of compensatory survival pathways
- Enhanced autophagy
- Heterogeneity in COPD-specific senescence signatures [26].
Importantly, COPD-related senescence differs from chronological aging. Cigarette smoke induces oxidative DNA damage, telomere-independent senescence, and chronic NF-κB activation, suggesting that COPD-specific molecular targets may exist beyond general aging pathways.
Emerging senolytics [2023–2025]
Recent work has identified new drug classes with senolytic potential. HSP90 inhibitors can destabilize survival proteins in senescent cells [27]. USP7 inhibitors interfere with deubiquitination pathways and promote apoptosis [26]. Mitochondria-targeting agents that exploit senescent-cell bioenergetic vulnerabilities are under development, with encouraging early findings [28]. Artificial intelligence-based drug discovery has also accelerated the identification of small molecules with senolytic activity, representing a rapidly growing field.
Senolytics vs. senomorphics – Translational considerations
While senolytics eliminate senescent cells, senomorphics such as metformin and rapamycin modulate SASP expression without inducing apoptosis. In COPD, where epithelial repair capacity is already compromised, complete removal of senescent cells may risk impairing tissue integrity [29].
Combination therapy [senolytic + senomorphic] could theoretically:
- Reduce senescent burden [senolytic effect]
- Suppress residual SASP signaling [senomorphic effect]
- Permit lower dosing of each agent
The comparative risk–benefit profile of these approaches is summarized in Table 1.
Comparative evaluation of senolytic agents in respiratory models
Although multiple senolytic agents demonstrate activity in experimental systems, their relative efficacy and translational feasibility differ substantially. Among currently studied agents, the Dasatinib–Quercetin combination shows consistent reduction in inflammatory infiltration and partial restoration of lung compliance in cigarette smoke–induced murine models. However, its efficacy appears dependent on intermittent dosing schedules and may vary based on disease severity.
Navitoclax demonstrates potent senolytic activity through BCL-2 family inhibition and produces robust clearance of senescent fibroblasts. Nevertheless, its translational feasibility in COPD is limited by dose-dependent thrombocytopenia resulting from BCL-XL inhibition in platelets [32].
Fisetin, in contrast, exhibits milder but safer senolytic activity, with additional antioxidant properties via Nrf2 activation. While its efficacy in COPD-specific models appears less dramatic than Navitoclax, its favorable safety profile enhances translational appeal.
FOXO4–DRI represents a highly specific mechanism-driven strategy targeting senescence survival signaling. However, limited pharmacokinetic data and peptide stability concerns restrict immediate clinical applicability.
Overall, BCL-2–targeting agents demonstrate higher potency but carry safety liabilities, whereas natural flavonoid-based senolytics offer improved tolerability but potentially lower efficacy. Future COPD-specific trials must balance senolytic potency with safety, particularly in elderly patients with comorbidities [33].
A comparative summary of the pharmacological characteristics, molecular targets, and limitations of senolytic and senomorphic agents relevant to COPD is presented in Table 2.
From a translational perspective, the developmental status of these agents varies considerably. Dasatinib–Quercetin and Fisetin have entered early-phase human studies in aging or non-COPD pulmonary conditions, whereas Navitoclax and HSP90 inhibitors are clinically evaluated primarily in oncology settings, with COPD evidence remaining preclinical. In contrast, metformin and rapamycin are already approved for other indications and possess well-characterized pharmacokinetic profiles, although their role in COPD is investigational. Most senolytics are administered orally, while peptide-based approaches such as FOXO4–DRI remain in preclinical development with delivery optimization [including inhalable formulations] under exploration. These distinctions highlight the gap between mechanistic promise and clinical feasibility in COPD [37] (Table 3).
Senolytics in COPD: Current evidence
Preclinical studies:. Most evidence for senolytics in respiratory disease stems from animal models. In cigarette smoke-exposed mice, periodic administration of senolytic agents reduces inflammatory cell infiltration, improves alveolar structure, and decreases elastase-induced tissue destruction [44]. Dasatinib–Quercetin and Navitoclax have both demonstrated reductions in pulmonary SASP markers, restoration of epithelial barrier integrity, and attenuation of airspace enlargement. Senolytic therapy also appears to benefit macrophage polarization and reduce oxidative stress within the lung microenvironment [45].
Early human data: Human studies remain scarce. Trials evaluating Dasatinib–Quercetin and Fisetin in idiopathic pulmonary fibrosis have shown reductions in senescence-related biomarkers, suggesting possible translatability to COPD [46-48]. However, human COPD-specific trials have not yet been completed, largely due to concerns surrounding toxicity, long-term safety, and lack of validated senescence biomarkers [49-51].
Drug delivery considerations: Systemic delivery may lead to off-target apoptosis and unwanted toxicity. Inhaled formulations of senolytics are being explored to achieve localized, high drug concentrations with reduced systemic exposure. Nanoparticle-based delivery systems may further enhance selective uptake by senescent lung cells [52].
Challenges and limitations
Despite promising preclinical data, several challenges impede clinical translation. Senolytics risk off-target apoptosis, particularly in cell populations with similar survival pathways. Navitoclax-associated thrombocytopenia highlights the importance of selective targeting [52]. Reliable biomarkers to quantify senescence burden in COPD patients are lacking. Without these, assessing therapeutic response becomes difficult [53]. Repeated dosing also raises concerns regarding tissue regeneration and immune function, as complete removal of senescent cells may not always be beneficial. Interpatient variability in senescence pathways suggests that personalized approaches may be required. Regulatory and ethical considerations remain unresolved as well [54].
Future directions
Future research may focus on inhalable senolytic formulations, which could reduce toxicity while enhancing local efficacy. Combination therapy approaches, such as pairing senolytics with anti-inflammatory drugs or CFTR modulators, may provide synergistic benefits for specific COPD phenotypes [53]. Integration of artificial intelligence may facilitate rapid discovery of selective senolytic molecules with improved safety profiles. Identification of biomarkers such as circulating SASP components or molecular imaging markers is likely to be central to patient selection and treatment monitoring [54]. Understanding how senescent immune cells influence COPD progression may open additional therapeutic avenues. Overall, senolytics represent an evolving and promising area of pharmacology.
Conclusion
Senolytics have emerged as a novel pharmacological strategy for addressing lung aging and the accumulation of senescent cells in COPD. Several agents have demonstrated promising preclinical benefits, particularly in reducing inflammation and improving lung structure. While human evidence remains limited and safety concerns must be resolved, senolytics represent a potential therapeutic advancement for COPD and related age-associated respiratory disorders.
Acknowledgment
The author gratefully acknowledges the Faculty of Pharmacy, Noble University, Junagadh, Gujarat, and Dr. Subhash Ayurveda Research Institute, Dr. Subhash University, for providing academic support and access to essential resources that contributed to the completion of this work. Their continued encouragement and guidance are sincerely appreciated.
References
- Barnes PJ. Cellular and molecular mechanisms of chronic obstructive pulmonary disease. Clin Chest Med. 2014;35(1):71–86. Available from: https://doi.org/10.1016/j.ccm.2013.10.004
- Tsuji T, Aoshiba K, Nagai A. Alveolar cell senescence in aging and chronic lung diseases. Respir Investig. 2012;50(1):30–37.
- Birch J, Passos JF. Targeting the SASP to combat ageing: Mitochondria as possible intracellular allies? Bioessays. 2017;39(5):1600235. Available from: https://doi.org/10.1002/bies.201600235
- Kirkland JL, Tchkonia T. Cellular senescence: A translational perspective. EBioMedicine. 2017;21:21–28. Available from: https://doi.org/10.1016/j.ebiom.2017.04.013
- Nyunoya T, Monick MM, Klingelhutz A, Yarovinsky TO, Cagley JR, Hunninghake GW. Cigarette smoke–induced cellular senescence in the lung. Am J Respir Cell Mol Biol. 2014;51(3):361–369. Available from: https://doi.org/10.1165/rcmb.2006-0169oc
- Sharpless NE, Sherr CJ. Forging a signature of in vivo senescence. Nat Rev Cancer. 2015;15(7):397–408. Available from: https://doi.org/10.1038/nrc3960
- Coppe JP, Desprez PY, Krtolica A, Campisi J. The senescence-associated secretory phenotype: The dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118. Available from: https://doi.org/10.1146/annurev-pathol-121808-102144
- Walters MS. Airway epithelial senescence in COPD. Am J Physiol Lung Cell Mol Physiol. 2014;306(5):L364–L370.
- Sohal SS. Epithelial and endothelial cell senescence in COPD. Chest. 2020;158(2):476–488. Available from: https://doi.org/10.1016/j.resinv.2016.11.006
- Zhu Y, Tchkonia T, Pirtskhalava T, Gower AC, Ding H, Giorgadze N, et al. The Achilles’ heel of senescent cells: Targeting anti-apoptotic pathways. Aging Cell. 2015;14(4):644–658. Available from: https://doi.org/10.1111/acel.12344
- Chilosi M, Carloni A. Cellular senescence and chronic lung disease. Curr Opin Pharmacol. 2021;56:1–9.
- Houssaini A. Senescence in pulmonary diseases. Eur Respir Rev. 2021;30(159):200261.
- Xu M, Pirtskhalava T, Farr JN, Weigand BM, Palmer AK, Weivoda MM, et al. Dasatinib and quercetin as senolytics. Nature Med. 2018;24(8):1246–1256.
- Schafer MJ. Senolytics improve lung function in aged mice. Aging Cell. 2017;16(5):973–977.
- Kirkland JL. Intermittent senolytic dosing. J Am Geriatr Soc. 2020;68(S2):S7–S13.
- Yousefzadeh MJ, Zhu Y, McGowan SJ, Angelini L, Fuhrmann-Stroissnigg H, Xu M, et al. Fisetin is a senotherapeutic compound. EBioMedicine. 2018;36:18–28. Available from: https://doi.org/10.1016/j.ebiom.2018.09.015
- Chen H. Fisetin prevents cigarette smoke–induced lung inflammation. J Inflamm Res. 2022;15:2671–84
- Yosef R. Navitoclax as a senolytic agent. Nat Commun. 2016;7:11190.
- Lehmann M. Senolytic therapy in lung fibrosis. Nat Commun. 2017;8:14532.
- Rudin CM. Navitoclax toxicity profile. J Clin Oncol. 2012;30(9):1002–1008.
- Baar MP. FOXO4–DRI peptide targets senescent cells. Cell. 2017;169(1):132–147.
- Parikh P. FOXO4 modulation in pulmonary senescence. Respir Res. 2019;20(1):254.
- Kulkarni AS, Gubbi S, Barzilai N. Metformin in aging biology. Cell Metab. 2020;32(1):15–25.
- Houssaini A. mTOR signaling in lung aging. Sci Transl Med. 2018;10(449):eaao4163.
- Fuhrmann-Stroissnigg H, et al. HSP90 inhibitors as senolytics. Cell Rep. 2017;21(1):199–208.
- Li J. USP7 inhibition promotes senolysis. Aging Cell. 2021;20(1):e13305.
- Chapman J. Mitochondrial-targeting senolytics. Geroscience. 2022;44(2):885–900.
- Yan J. AI-driven identification of senolytics. Nat Biotechnol. 2023;41(2):216–225.
- Chuang HC. Senolytics in cigarette smoke–induced models. Environ Pollut. 2022;292:118369.
- Birukova A. Senolytic modulation of airspace enlargement. Am J Respir Crit Care Med. 2023;207(4):449–460.
- Lin W. Macrophage senescence in COPD models. J Transl Med. 2021;19(1):236.
- Justice JN, Nambiar AM, Tchkonia T, LeBrasseur NK, Pascual R, Hashmi SK, et al. Senolytics in pulmonary fibrosis patients. EBioMedicine. 2019;40:554–563. Available from: https://doi.org/10.1016/j.ebiom.2018.12.052
- Mehdizadeh M. Clinical challenges of senolytics. Drugs Aging. 2022;39(2):127–141.
- Li X. Inhalable senolytics for lung diseases. Adv Drug Deliv Rev. 2023;198:114903.
- Ramanathan S. Nanocarrier-based senolytics. Int J Pharm. 2024;635:122897.
- Ashkenazi A. Mechanisms of Navitoclax toxicity. Cell Death Dis. 2020;11(9):712.
- Narita M. Senescence biomarkers in clinical research. Nat Rev Mol Cell Biol. 2019;20(11):671–684.
- Childs BG. Risks of senolytic therapy. Nat Med. 2015;21(12):1424–1435.
- Vasto S. Ethics of senolytic treatment. Aging Clin Exp Res. 2023;35:1607–1614.
- Fini MA. Combination senolytic strategies. J Mol Med. 2024;102:1501–1512.
- Wang C. Biomarkers for senescence-targeted therapy. Trends Pharmacol Sci. 2024;45(3):217–229.
- Zhu Y, Tchkonia T, Fuhrmann-Stroissnigg H, Dai HM, Ling YY, Stout MB, et al. Identification of a novel senolytic agent targeting the BCL-2 family of anti-apoptotic factors. Aging Cell. 2016;15(3):428–435. Available from: https://doi.org/10.1111/acel.12445
- Kirkland JL, Tchkonia T. Senolytic drugs: From discovery to translation. J Intern Med. 2020;288(5):518–536. Available from: https://doi.org/10.1111/joim.13141
- Baar MP, Brandt RMC, Putavet DA, Klein JD, Derks KWJ, Bourgeois BRM, et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell. 2017;169(1):132–147.e16. Available from: https://doi.org/10.1016/j.cell.2017.02.031
- Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of cellular senescence. Trends Cell Biol. 2018;28(6):436–453. Available from: https://doi.org/10.1016/j.tcb.2018.02.001
- Ito K, Barnes PJ. COPD is a disease of accelerated lung aging. Chest. 2009;135(1):173–180. Available from: https://doi.org/10.1378/chest.08-1419
- Tsuji T, Aoshiba K, Nagai A. Alveolar cell senescence in aging and chronic lung diseases. Respir Investig. 2012;50(1):30–37. Available from: https://doi.org/10.1164/rccm.200509-1374oc
- Childs BG, Durik M, Baker DJ, van Deursen JM. Cellular senescence in aging and age-related disease: From mechanisms to therapy. Nat Med. 2015;21(12):1424–1435. Available from: https://doi.org/10.1038/nm.4000
- Niedernhofer LJ, Kirkland JL, Ladiges W. Molecular pathology endpoints useful for aging studies. Nat Rev Drug Discov. 2018;17(5):377–377.
- Fini MA, Monzon ME, Campos M, Declerck YA. Combination senolytic strategies in chronic lung diseases. J Mol Med. 2024;102(11):1501–1512.
- Justice JN, Nambiar AM, Tchkonia T, LeBrasseur NK, Pascual R, Hashmi SK, et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human pilot study. EBioMedicine. 2019;40:554–563. Available from: https://doi.org/10.1016/j.ebiom.2018.12.052
- Mehdizadeh M, Aguilar M, Thorin E. Clinical challenges and translational considerations of senolytic therapy. Drugs Aging. 2022;39(2):127–141.
- Li X, Wu Y, Chen J, Zhang M, Gong L. Inhalable delivery systems for senolytic therapy in lung diseases. Adv Drug Deliv Rev. 2023;198:114903.
- Ramanathan S, Subramanian R, Iyer AKV. Nanocarrier-based pulmonary delivery of senotherapeutics: Emerging strategies. Int J Pharm. 2024;635:122897.


 Save to Mendeley
Save to Mendeley

